All About Corrosion and Its Types
Introduction to Corrosion
Corrosion is a natural electrochemical process that causes the deterioration of materials, especially metals, through chemical or electrochemical reactions with their environment. It is a significant challenge for industries such as construction, automotive, oil and gas, and maritime operations. Understanding corrosion and its various types is essential to developing effective strategies to prevent material degradation, reduce maintenance costs, and ensure the longevity and safety of structures and equipment.
The Science Behind Corrosion
Corrosion involves the transfer of electrons from a metal to a surrounding medium. The metal acts as an anode, losing electrons and undergoing oxidation, while another material or area serves as the cathode, gaining electrons. Moisture, oxygen, and pollutants accelerate these reactions, forming oxides or other compounds that damage the metal surface.
Classification of Corrosion Types
Corrosion can manifest in several distinct forms, each with unique characteristics, causes, and consequences.
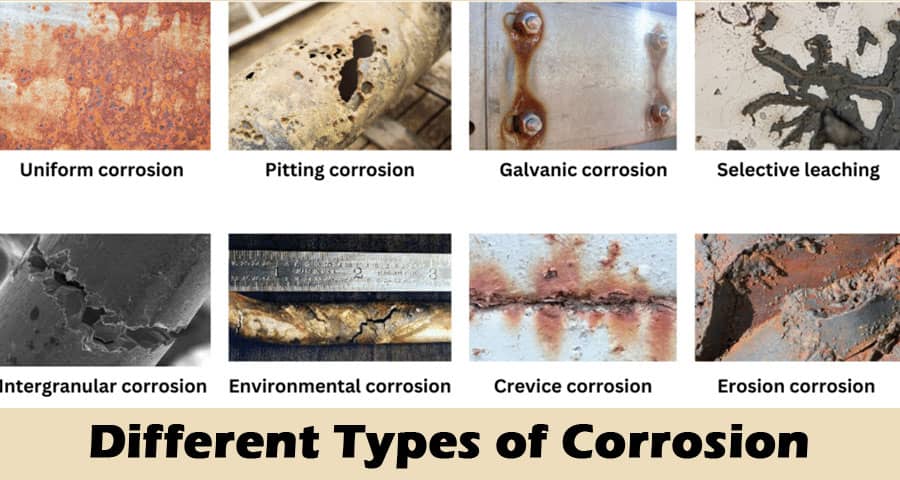
1. Uniform Corrosion
Uniform corrosion is the most common and widespread form, characterized by an even metal loss across the entire exposed surface. This type is usually predictable and can be managed by applying protective coatings, using corrosion-resistant alloys, or implementing cathodic protection systems. Uniform corrosion occurs in environments with consistent exposure to corrosive agents, such as open steel structures in humid or marine settings.
2. Galvanic Corrosion
Galvanic corrosion arises when two dissimilar metals come into electrical contact in the presence of an electrolyte, such as water. The more active metal (anode) corrodes faster, while the nobler metal (cathode) is protected. This often happens in marine structures, pipelines, or equipment with mixed metal assemblies. Preventative measures include using metals with similar electrochemical potentials, isolating metals with insulating gaskets, or applying protective coatings.

3. Pitting Corrosion
Pitting corrosion is a localized form that leads to small but deep cavities or pits on metal surfaces. Chloride ions, often from saline environments, are primary culprits in initiating pits on stainless steels and aluminum alloys. Pitting is especially dangerous because it can cause sudden failures with minimal overall material loss, making early detection and preventive maintenance critical.
4. Crevice Corrosion
Crevice corrosion develops in confined spaces where stagnant solutions can accumulate, such as under gaskets, washers, lap joints, or deposits. The restricted exchange of oxygen leads to acidic microenvironments and aggressive localized attack. Crevice corrosion is common in marine equipment, tanks, and bolted structures. Design improvements, sealant use, and regular cleaning help mitigate this form of corrosion.
5. Intergranular Corrosion
Intergranular corrosion affects the grain boundaries of metals, typically due to the precipitation of carbides or other phases that cause chromium depletion in stainless steels. This weakens the material's structural integrity, often leading to catastrophic failures. Susceptibility can be reduced by using low-carbon or stabilized grades of stainless steel and applying proper heat treatments to dissolve harmful precipitates.
6. Stress Corrosion Cracking (SCC)
SCC is a dangerous synergy of tensile stress and corrosive environments, causing cracks to form and propagate without significant plastic deformation. Chloride-induced SCC in austenitic stainless steels is a common example. SCC can lead to sudden, unexpected failures in pipelines, pressure vessels, and components under constant load. Mitigation involves reducing tensile stresses, controlling environmental exposure, and selecting resistant materials.
7. Erosion Corrosion
Erosion corrosion occurs when a high-velocity fluid stream causes mechanical wear in combination with corrosion, leading to accelerated material loss. It often affects pipe bends, pump impellers, and heat exchanger tubes exposed to slurries or turbulent flow. Solutions include using erosion-resistant materials, optimizing flow rates, and redesigning equipment to minimize turbulent zones.
8. Microbiologically Influenced Corrosion (MIC)
MIC involves the activity of microorganisms, such as sulfate-reducing bacteria, that create corrosive environments through their metabolic processes. MIC is prevalent in water distribution systems, fuel storage tanks, and offshore platforms. Effective control includes biocides, regular system cleaning, and maintaining dry, oxygen-free conditions where feasible.

9. Hydrogen Embrittlement
Hydrogen embrittlement results from hydrogen atoms diffusing into metals, causing them to become brittle and crack under stress. This type is common in high-strength steels exposed to cathodic protection, pickling, or electroplating processes. Preventive strategies include controlling exposure to hydrogen sources and using tempered materials.
Corrosion Prevention and Protection Methods
Effective corrosion control is critical to ensuring the durability of metallic components. We must adopt a combination of preventive measures to protect against different types of corrosion:
- Protective Coatings: Application of paints, powder coatings, or metallic coatings like galvanizing creates a barrier between the metal and corrosive agents.
- Cathodic Protection: This technique uses sacrificial anodes or impressed current systems to shift the potential of the metal to a non-corrosive state.
- Material Selection: Choosing corrosion-resistant alloys, stainless steels, or non-metallic alternatives based on environmental conditions greatly reduces susceptibility.
- Environmental Control: Reducing moisture, pollutants, or aggressive chemicals in the operating environment minimizes corrosion risks.
- Design Considerations: Avoiding crevices, ensuring good drainage, and designing for easy maintenance can significantly lower corrosion likelihood.
- Regular Inspection and Maintenance: Scheduled inspections using techniques like ultrasonic testing, radiography, or corrosion coupons help detect early signs of degradation.
Real-World Impacts of Corrosion
Corrosion poses severe threats to safety, economy, and the environment. Infrastructure failures, such as bridge collapses or pipeline ruptures, have led to injuries, fatalities, and massive financial losses. The global cost of corrosion is estimated to exceed 3% of the world's GDP, highlighting the importance of effective corrosion management strategies.
Industries that heavily invest in corrosion control, such as oil and gas, aviation, and maritime operations, benefit from reduced downtime, extended equipment life, and improved safety records. Moreover, preventing corrosion contributes to environmental protection by minimizing leaks and spills of hazardous substances.
Advanced Technologies in Corrosion Monitoring
Recent advancements in corrosion monitoring technologies provide real-time data to predict and prevent failures. Electrochemical sensors, fiber optic systems, and wireless monitoring networks allow for continuous assessment of corrosion rates in harsh environments. These technologies enable proactive maintenance, optimizing asset management and reducing unplanned shutdowns.
Conclusion
Understanding the types of corrosion and implementing comprehensive protection measures are vital for the longevity of metallic structures and components. By leveraging advanced materials, modern design practices, and cutting-edge monitoring systems, industries can significantly reduce the risks and costs associated with corrosion, ensuring safer and more sustainable operations.
Please view the following short video for All About Corrosion and Its Types
